Antidepressant and Anxiolytic Effects of Medicinal Cannabis Use in an Observational Trial
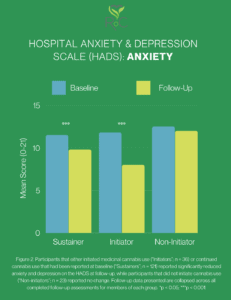
Understanding RoC’s latest published research on cannabinoid therapy and anxiety/depression. Between April 2016 and July 2020, 538 participants were enrolled in an observational research study between Realm of Caring
